

instituto de biologia molecular e celular | institute for molecular and cell biology
Research Interests
The Neurodegeneration Group is mainly focused on the study of neuronal cytoskeleton remodeling in protein aggregate-induced neurodegenerative disorders. We are very interested in defining common pathogenic mechanisms among several neurodegenerative conditions, specifically by determining cytoskeleton alterations induced by different proteins aggregates. The establishment of overlapping cellular targets across different neurodegenerative disorders will be crucial for designing common therapeutic interventions.
We are currently focusing in Familial Amyloid Polyneuropathy (FAP), Parkinson´s Disease (PD) and Alzheimer´s Disease (AD). We use primary neuronal cultures treated with protein aggregates, combined with cell biology methodologies and live-cell imaging, to characterize their effect on the cytoskeleton organization of the cell. Additionally, cytoskeleton alterations are validated in mouse models of disease and patient-derived samples.
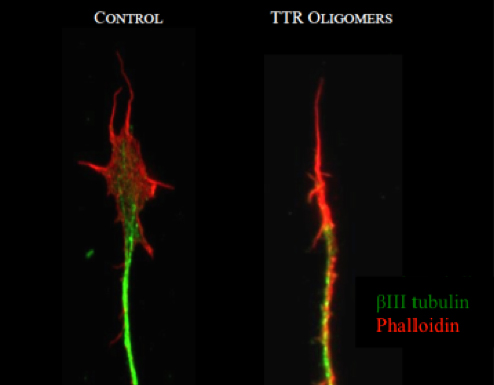
IMAGE: Cytoskeleton organization of the growth cone of DRG neurons untreated (CONTROL) or treated with TTR oligomers.
Research Projects
In the context of FAP, a neurodegenerative disorder characterized by the deposition of Transthyretin (TTR) aggregates particular in the peripheral nervous system, we obtained data suggesting a disruption of the actin cytoskeleton upon mutant TTR deposition. We are currently addressing the molecular mechanisms underlying TTR-induced actin cytoskeleton alterations.
In PD, a disease characterized by the accumulation of -synuclein cytoplasmic inclusions in the substantia nigra, we demonstrated that α-synuclein oligomers induce the formation of cofilin-actin rods in hippocampal neurons. Future work will determine whether α-synuclein-induced rods underlie the cognitive dysfunction observed in PD patients.
In AD, we demonstrated that TTR cleavage of Aβ underlies the neuroprotective effect of TTR in the disease (“Transthyretin neuroprotection in Alzheimer´s disease is dependent on proteolysis”, In preparation).
Publications
Eira J, Silva CS, Sousa MM, Liz MA (2016). The cytoskeleton as a novel therapeutic target for old neurodegenerative disorders. Progress in Neurobiology 141:61-82.
Liz MA, Mar FM, Santos TE, Pimentel HI, Marques AM, Morgado MM et al (2014). Neuronal deletion of GSK3β increases MT speed in the growth cone and enhances axon regeneration via CRMP-2 and independently of MAP1B and CLASP2. BMC Biology 12:47.
Liz MA, Leite SC, Juliano L, Saraiva MJ, Damas AM, Bur D et al (2012). Transthyretin is a metallopeptidase with an inducible active site. Biochemical Journal 443:769-78.
Miranda CO, Teixeira CA, Liz MA, Sousa VF, Franquinho F, Forte G, di Nardo P, Pinto-do-Ó P and Sousa MM (2011). Systemic delivery of bone marrow-derived mesenchymal stromal cells diminishes neuropathology in a mouse model of Krabbe's disease. Stem Cells 29: 1738-51.
Liz MA, Fleming CE, Nunes AF, Almeida MR, Mar FM, Choe Y, Craik CS, Powers JC, Bogyo M and Sousa MM (2009). Substrate specificity of transthyretin: identification of natural substrates in the nervous system. Biochemical Journal 419: 467-74.

Magalhães, Joana Miguel
jmmagalhaes@i3s.up.pt
Faria da Eira, Jessica
jessica.eira@ibmc.up.pt
Isabel Silva, Marina
marina.silva@i3s.up.pt
Home | Site Map | Contacts | Credits | Privacy & Cookies | WHISTLEBLOWER CHANNEL | Intranet | Social Networks |
